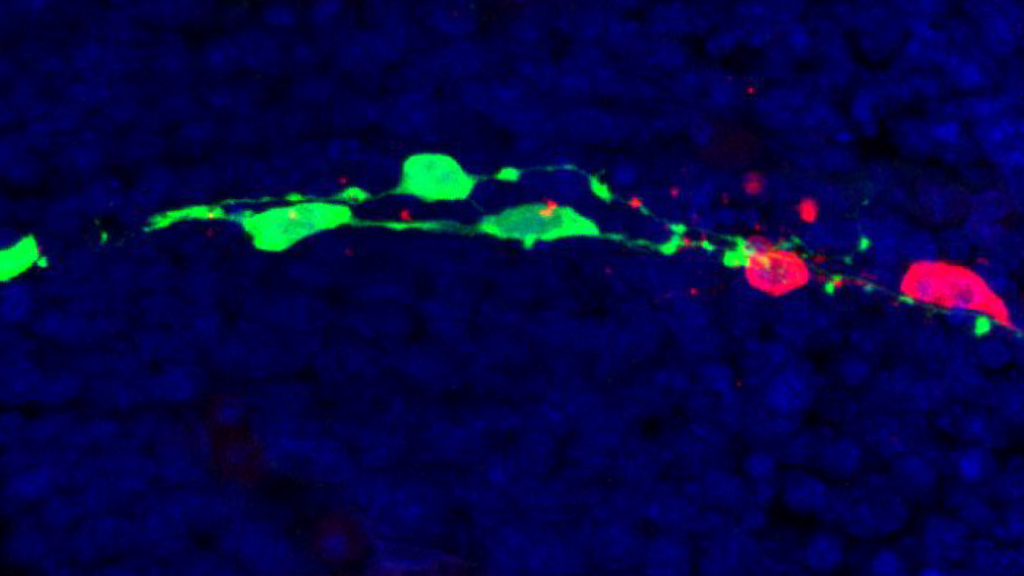
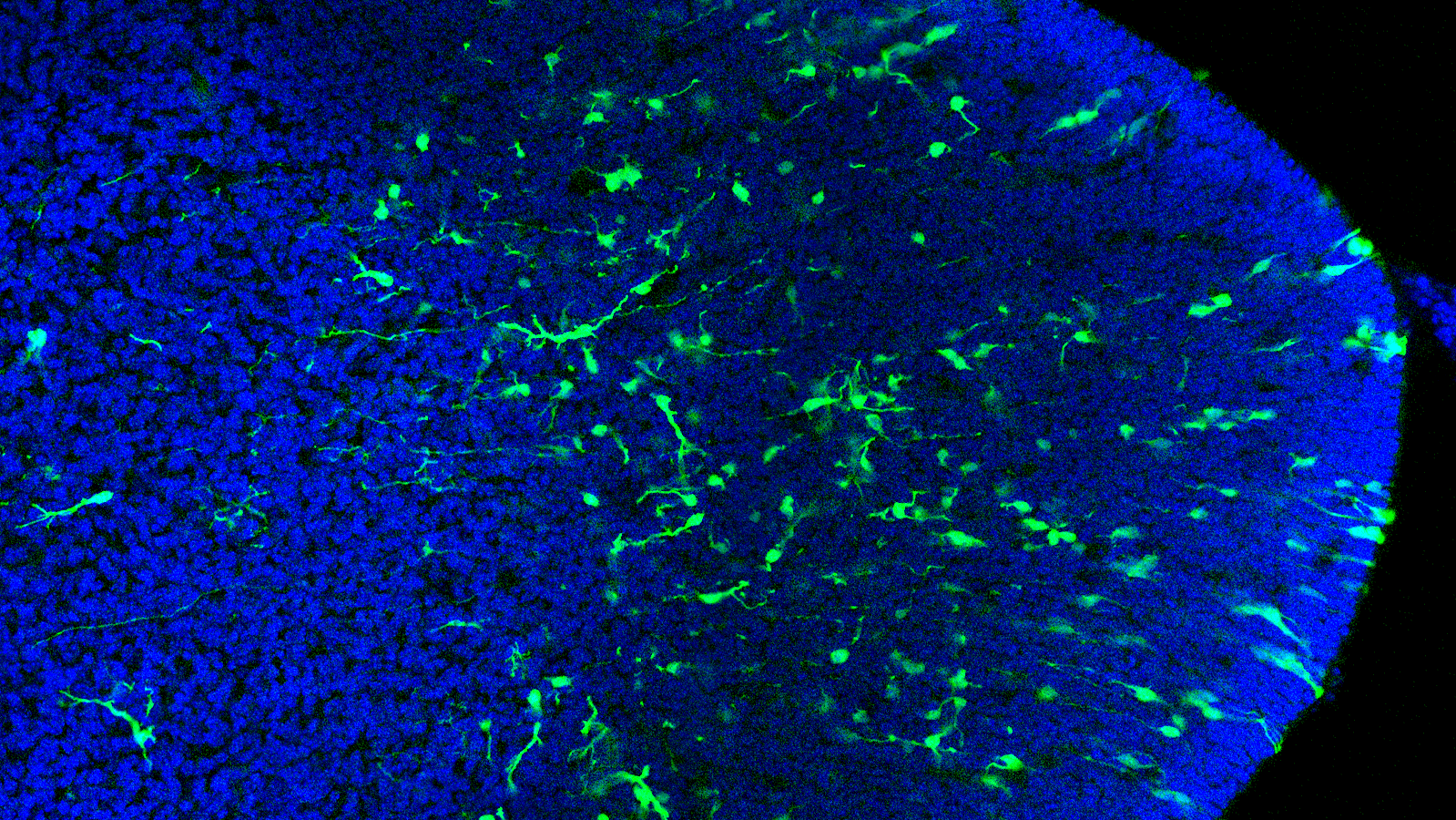
VISION
We want to understand how the human brain builds its inhibitory neurons — and what happens when this process goes wrong in neurodevelopmental and psychiatric disease. Inhibitory neurons are essential for the balance of brain circuits, and their dysfunction recurs across autism, schizophrenia, and epilepsy. Our laboratory studies how the diversity of these neurons emerges during early development, and how mutations in disease-associated genes alter this process at its origin.
RECENT HIGHLIGHTS
Temporal control of progenitor competence shapes GABAergic neuron maturation
Bright et al., Nature Neuroscience (2025).
Combining single-cell multi-omics with birthdating, we show that the timing of neurogenesis controls a progenitor’s capacity to mature into a functional neuron, but does not constrain cell-type diversity. NFIB-driven chromatin priming emerges as a key mechanism through which intrinsic developmental programs interact with the cellular environment.
Spatial enhancer activation influences inhibitory neuron identity
Dvoretskova et al., Nature Neuroscience (2024).
We introduce an in vivo platform that combines clonal lineage tracing with CRISPR perturbation sequencing, allowing us to dissect the function of disease-linked transcription factors at clonal resolution. The study reveals that MEIS2, cooperating with DLX5 and antagonized by LHX6, activates projection-neuron enhancers — and that Meis2 mutations associated with neurodevelopmental disorders disrupt this regulation.
Single-cell delineation of lineage and genetic identity in the mouse brain
Bandler, Vitali et al., Nature (2022).
Using a massively parallel progenitor-tagging strategy combined with single-cell transcriptomics, we developed a framework to simultaneously resolve clonal relationships and molecular identity in the developing mouse brain. The approach is now widely used for lineage analysis in development and disease models.
Developmental diversification of cortical inhibitory interneurons
Mayer et al., Nature (2018).
Among the first studies to apply single-cell RNA sequencing across an extended developmental time course, this work showed that adult interneuron identities are already detectable in the embryonic forebrain shortly after cell-cycle exit. It identified MEF2C — a gene genetically linked to schizophrenia and other neurodevelopmental disorders — as a key regulator of parvalbumin interneuron specification.
RESEARCH AREAS
1. Tracing inhibitory neuron lineages from embryo to adult.
We use barcoded lineage tracing in combination with single-cell transcriptomics and chromatin profiling to follow how individual progenitors in the embryonic forebrain give rise to the full diversity of GABAergic neurons. We ask when and how fate decisions are made, and which molecular signatures predict which adult neuronal identity.
2. How transcription factors specify neuronal identity at the molecular level.
Inhibitory neuron identity is established through cooperative interactions between transcription factors and chromatin regulators at enhancers — short stretches of DNA that switch genes on at the right time and place. Using ChIP-seq, CUT&RUN, co-immunoprecipitation coupled to mass spectrometry, enhancer pull-downs, reporter assays, and engineered protein mutations, we dissect how key regulators such as MEIS2, DLX5, LHX6, SP9, and NFI-family factors (NFIA/B/X) work together to establish lineage-specific enhancer activity.
3. Neuropsychiatric risk genes and inhibitory neuron development.
Many high-confidence risk genes for autism spectrum disorder and schizophrenia — including chromatin regulators and transcription factors — act within early inhibitory neuron lineages. Using in vivo CRISPR perturbation at clonal resolution, we systematically test how disruption of these genes alters developmental trajectories, enhancer activity, and the final balance of neuronal cell types. This work links genetic risk directly to defined cellular and molecular phenotypes.
4. From development to circuits.
Together with computational modeling and barcoded monosynaptic rabies tracing (RabV-seq), we connect developmental perturbations to circuit-level outcomes, asking how altered inhibitory neuron development reshapes the wiring of neural circuits.
5. Methods for clonally resolved perturbation biology.
We have developed methods that combine genetic barcoding, in vivo CRISPR perturbation, and single-cell multi-omics to perform parallel functional screens at clonal resolution in the developing brain. Ongoing developments include machine-learning approaches for lineage reconstruction and the extension of these tools from mouse models to human iPSC-based systems and brain organoids.
CURRENTLY EXPLORING
– How does SP9 balance D1 versus D2 neuron production?
Neuronal diversity arises when common progenitors deploy distinct gene expression programs to generate specialized cell types — but how transcription factors are directed to promote one developmental fate while restraining another remains poorly understood. We are studying how the transcription factor SP9 controls the balance between D1 and D2 medium spiny neuron identities during mouse basal ganglia development. Using clonally resolved single-cell perturbation in vivo, we find that loss of Sp9 shifts cell production away from D2 neurons and toward D1 neurons, identifying SP9 as a determinant of subtype allocation. Mechanistically, SP9 directly binds canonical SP-family motifs at D2 fate genes, but can also be recruited to DLX-bound homeobox motifs at distal regulatory elements, where SP9–DLX interaction attenuates enhancer activity. The relative levels of SP9 and DLX appear to influence which class of binding sites is occupied — suggesting partner-dependent control of transcription factor binding as a mechanism for tuning neuronal subtype output, with implications for how disrupted developmental gene regulation may contribute to neuropsychiatric disease.
– How do disease-risk genes shape developing neural circuits?
Using parallelized in vivo CRISPR perturbations of high-confidence autism risk genes, we ask which genes disrupt inhibitory neuron development and which converge on shared mechanisms.
– Can clonal lineage tracing in human brain organoids recapitulate the regulatory logic identified in mouse?
We are extending our perturbation and lineage-tracing platform to human iPSC and organoid systems.
– Are early developmental defects reversible?
Using temporally controlled gene-perturbation systems, we test whether developmental alterations caused by high-risk neuropsychiatric genes can be rescued after their onset.
TRANSLATIONAL RELEVANCE
Inhibitory neuron dysfunction recurs across autism spectrum disorder, schizophrenia, and epilepsy. By uncovering the regulatory mechanisms that build these neurons during embryonic development, our work links genetic risk to the molecular events that go awry in disease — a foundation for understanding, and ultimately addressing, neurodevelopmental disorders.

COLLABORATIVE RESEARCH
The MPI for Biological Intelligence is part of a vibrant Munich research campus, with close interactions across the MPIs of Biochemistry and Psychiatry, Ludwig-Maximilian University, the Technical University of Munich, and Helmholtz Munich. We actively participate in:
Graduate School of Systemic Neuroscience (GSN)
International Max Planck Research Schools: IMPRIS-BI and IMPRS-TP
Funding
Inhibitory Neuron Development Lab
Research at the Max Planck Institute for Biological Intelligence